Question 80
(Multiple Choice)
The solutions in the arms of a U-tube are separated at the bottom of the tube by a selectively permeable membrane. The membrane is permeable to sodium chloride but not to glucose. Side A is filled with a solution of 0.4 M glucose and 0.5 M sodium chloride (NaCl), and side B is filled with a solution containing 0.8 M glucose and 0.4 M sodium chloride. Initially, the volume in both arms is the same. Refer to the figure to answer the following questions.
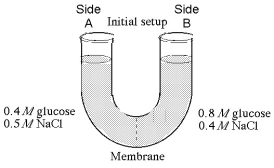
-If you examine side A after three days, you should find
A) a decrease in the concentration of NaCl and glucose and an increase in the water level.
B) a decrease in the concentration of NaCl, an increase in water level, and no change in the concentration of glucose.
C) no net change in the system.
D) a decrease in the concentration of NaCl and a decrease in the water level.
E) no change in the concentration of NaCl and glucose and an increase in the water level.
Answer